Peptides that accelerate bone and fracture healing: evidence for research use
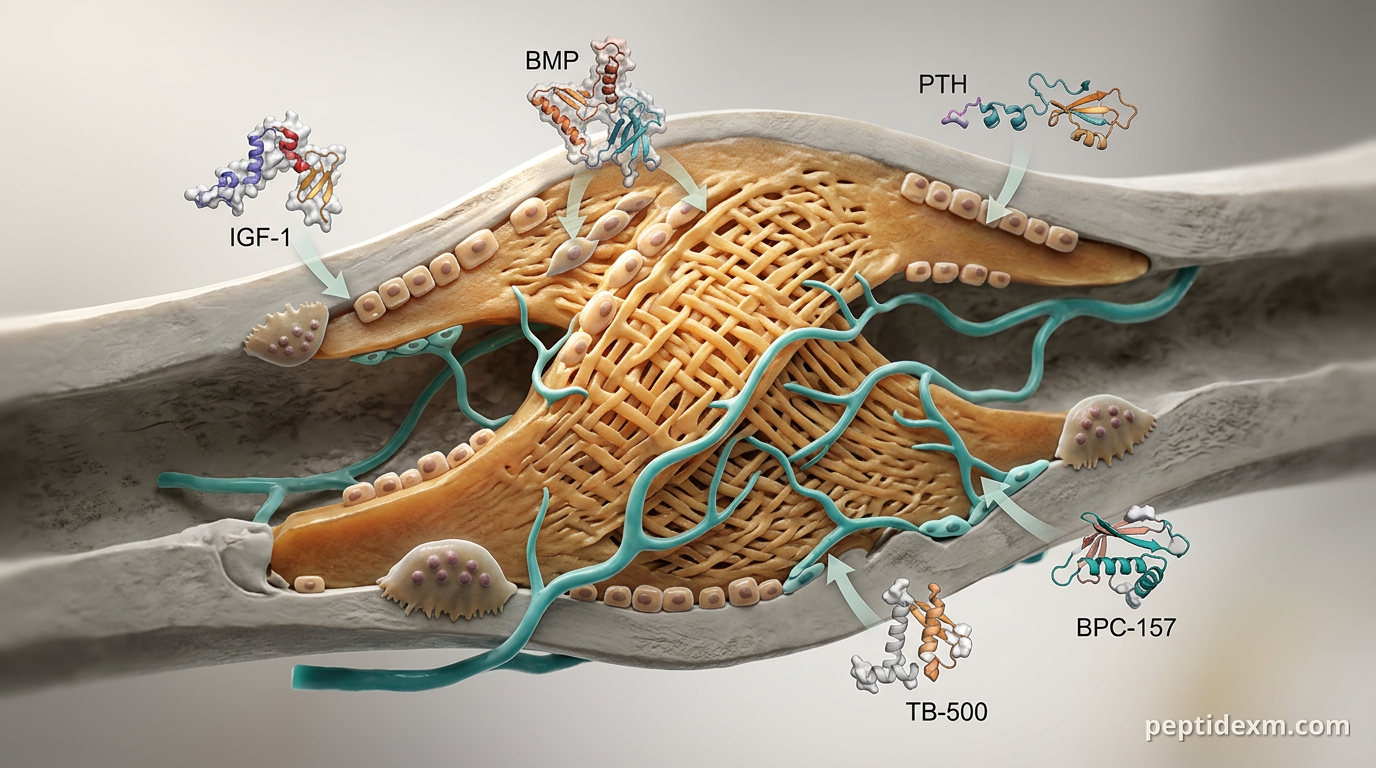
If you work with fracture models, you want agents that actually move the needle on callus formation, mineralization, and biomechanical strength. Several peptides look promising in preclinical studies; they act on osteogenesis, angiogenesis, inflammation, or the growth-hormone axis. This post summarizes the candidates, what the evidence actually shows in lab models, and practical design and safety notes for research use only.
Which peptide classes show the strongest preclinical signal? Across rodent and large-animal fracture models, three mechanistic buckets consistently emerge: direct osteoanabolic factors, pro-regenerative peptides that modulate inflammation and angiogenesis, and growth-hormone/IGF axis modulators. Each bucket contains multiple candidates and different experimental trade-offs.
Direct osteoanabolic: BMP family members and PTH fragments (widely studied clinically, but not sold here) drive osteoblast differentiation. Pro-regenerative/repair peptides: agents that boost angiogenesis, cell migration, or reduce excessive inflammation—examples include Thymosin Beta-4 (TB-500) and BPC-157. GH/IGF axis and muscle–bone modulators: IGF-1 and IGF-1 analogs increase matrix production and bone formation; follistatin family peptides can alter loading biology via muscle effects.
Note: this article stays strictly in the research domain. It doesn't recommend human use or dosing.
Key candidates and what the data actually show Below I summarize peptides that researchers commonly test for bone or fracture healing, and what endpoints improved in preclinical work. I include a quick practical note on each.
BPC-157 (stable gastric pentadecapeptide) BPC-157 comes up a lot in soft-tissue and musculoskeletal repair literature. In bone-focused studies it’s been associated with faster bridging of cortical defects and improved early callus organization in rodent models. The suggested mechanisms include modulation of local growth factor expression, enhancement of angiogenesis, and suppression of excessive inflammatory signaling. Most work is preclinical and sometimes uses local application into the defect or systemic delivery in animals.
IGF-1 and IGF-1 analogs (IGF-1 LR3) IGF-1 is central to osteoblast proliferation and matrix synthesis. In fracture models, IGF-1 increases callus size, mineral deposition, and mechanical strength when delivered locally or systemically in animals. Long-acting analogs like IGF-1 LR3 prolong bioactivity in circulation and in scaffolds, which can be useful for studies that require sustained signaling rather than multiple administrations.
Thymosin Beta-4 (TB-500) TB-500 promotes cell migration, angiogenesis, and cytoskeletal remodeling. In bone models it tends to improve vascular ingrowth into the fracture site and supports earlier remodeling in histology. Its value is often as a complement to osteoinductive factors—better blood supply means cells and nutrients reach the callus sooner.
Follistatin (FST344) and myostatin pathway modulators Follistatin neutralizes myostatin and related TGF-β ligands. By increasing local muscle mass or altering mechanical loading, follistatin-derived peptides can have secondary positive effects on bone healing in load-bearing models. Expect indirect effects: bigger muscle, altered strain patterns, and downstream increases in bone formation markers.
GH secretagogues and related peptides Peptides that act on the GH axis—GHRH analogs, ghrelin mimetics, or GH secretagogues—change circulating IGF-1 and growth-related signaling. In animal fracture studies, these agents can increase systemic anabolic drive and improve healing metrics, though results vary with age and baseline endocrine status of the animals.
GH/IGF combinatory strategies and BMP adjuncts Many studies show the biggest gains when an osteoinductive factor (BMP or PTH fragment) is combined with pro-regenerative peptides that support angiogenesis and inflammation resolution. Combination approaches often produce larger callus and higher mechanical strength than single agents in the same model.
Mechanisms you should measure Picking a peptide is one thing. Showing how it works is another. If you want mechanistic data, prioritize a small panel of cellular and tissue readouts rather than only radiology.
Micro-CT: callus volume, bone volume/total volume (BV/TV), trabecular thickness—standard quantitative backbone. Biomechanics: three-point bending or torsion testing for stiffness and ultimate load—these are the outcomes that matter for functional repair in animals. Histology: histomorphometry for endochondral vs. intramembranous bone, TRAP staining for osteoclasts, Safranin O for cartilage within callus. Molecular markers: RUNX2, osterix, alkaline phosphatase, osteocalcin for osteoblast activity; VEGF and CD31 for angiogenesis; inflammatory cytokines at early time points. Time course: sample multiple timepoints—early inflammation (days 3–7), soft callus (days 7–14), mineralization (weeks 2–4), and remodeling (weeks 6+ depending on species).
Delivery form, dosing frequency, and local vs systemic trade-offs How you deliver a peptide can be as important as which peptide you use. Short peptides clear fast. That’s why most successful preclinical protocols pair them with a delivery system or use analogs with longer half-lives.
Local delivery into the defect: concentrates the peptide at the repair site, reduces systemic exposure, and often needs lower total peptide mass. Common carriers: collagen sponges, hydrogels, PLGA microspheres, and calcium phosphate cement. Systemic dosing: simpler to administer for repeated experiments but requires higher doses and raises off-target concerns. Useful when the mechanism is endocrine (IGF-axis) rather than strictly local. Sustained-release strategies: encapsulation in particles, peptide–polymer conjugates, or linking to matrices preserves activity for days to weeks and reduces dosage frequency. Timing: early pro-angiogenic/inflammation-modulating therapy is often most effective when given within the first week post-fracture; anabolic signals for mineralization are more impactful during weeks 1–4 in rodent models.
Experimental design and controls that make results believable Bone repair studies are noisy. Design choices determine whether you find a real effect or false leads.
Use an established fracture model with reproducible fixation: stabilized femoral or tibial fracture models reduce variability in mechanical environment, which is a major confounder. Include both radiographic and mechanical endpoints. Increased bone volume without improved mechanical strength is an incomplete outcome. Run a dose-ranging pilot for efficacy and safety. If you use sustained-release matrices, characterize release kinetics in vitro before in vivo work. Timepoint multiplicity. If you only measure end-of-study, you miss the dynamics—when angiogenesis peaked, when the cartilage-to-bone transition happened, etc. Appropriate biological controls: vehicle, carrier-only, and a positive control (if ethical and available) such as a clinically validated osteoinductive factor in your model. Blinding and randomization. Histology and biomechanical testing should be blinded to treatment arms.
Safety, off-target effects, and translational challenges Peptides can sound specific, but they have pitfalls. Below are common safety and translational issues encountered in preclinical bone-repair work.
Ectopic bone formation: potent osteoinductive signals, especially BMPs, can induce bone where you don’t want it. Local containment and dose control are key. Systemic endocrine effects: GH/IGF axis modulation affects tissues systemically. Monitor body weight, glucose homeostasis markers, and organ histology in longer studies. Immunogenicity: repeated dosing with peptide analogs can provoke antibodies in some species—measure anti-peptide titers if you plan chronic administration. Tumor biology: growth-promoting signals theoretically risk stimulating neoplastic growth. Screen for proliferative changes in off-target tissues if your peptide increases mitogenic signaling. Reproducibility: batch-to-batch peptide purity, aggregation during storage, and carrier inconsistencies will confound results. Use analytical QC and stability checks.
Practical lab notes and common pitfalls A few bench-level tips save time and reduce ambiguous outcomes.
Verify peptide identity and purity with HPLC and mass spec before in vivo use. Low-level contaminants can change immune responses in small animals. Stability: some peptides degrade rapidly in aqueous solution. Prepare aliquots, store frozen, and avoid multiple freeze–thaw cycles. Carrier interactions: test for peptide binding to scaffold materials in vitro; strong binding can reduce bioavailability. Batch size: make sure all animals in a cohort receive peptide from the same manufacturing batch to limit variability. Document kinetics: pair any efficacy readout with pharmacokinetic or release-kinetic data so you can link exposure to effect.
Peptide approaches for fracture healing are an active area of preclinical research. BPC-157 and IGF-1 analogs show solid evidence for accelerating early repair and improving mechanical outcomes in animal studies; TB-500 and follistatin-based approaches help by enhancing angiogenesis and modifying the muscle–bone environment. Delivery form, timing, and robust outcome measures matter more than picking a single "magic" peptide.
For research use only; this post does not endorse human administration or clinical treatment. If you want references or a suggested study outline tailored to your model (rat femoral callus vs. rabbit critical-size defect), tell me your species and fixation method and I’ll sketch a protocol framework.