Peptides for Skin Glow, Nail Strength, and Beard
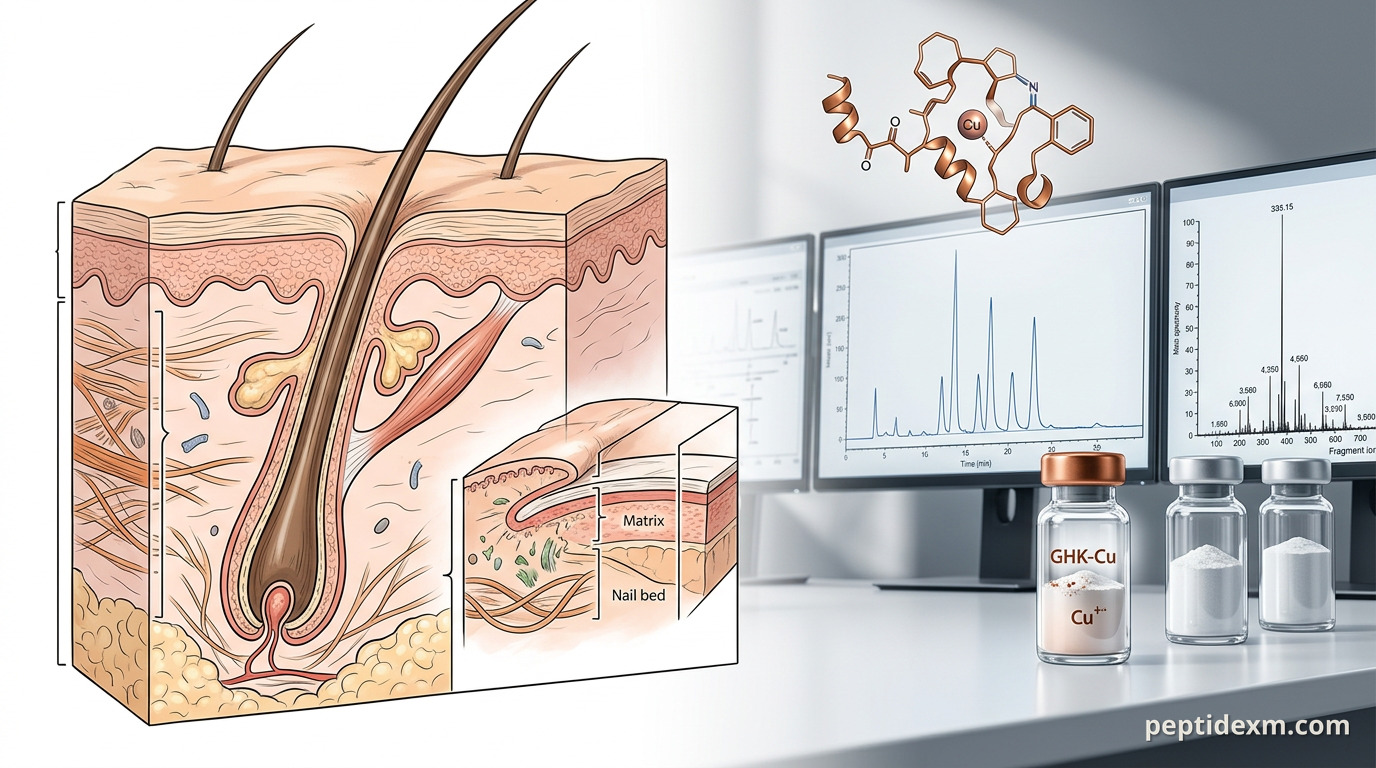
If you work with cosmetic or integumentary biology in a lab, peptides are one of the cleanest tools to probe repair, extracellular matrix dynamics, and follicle behaviour. This guide organizes the peptides people actually use in skin, nail, and beard research, explains plausible mechanisms, and gives practical notes on models, assays, formulation, and quality control. Research-only framing throughout—no human-use advice.
Why researchers look to peptides for skin, nails and facial hair
Peptides are small and modular. That makes them easier to synthesise, characterise, and modify than full proteins. In skin and appendage research, you typically want to alter one or more of these processes: collagen and elastin production, matrix metalloproteinase (MMP) activity, keratinocyte proliferation and differentiation, angiogenesis, or local inflammation. Peptides hit those targets directly or mimic short stretches of endogenous signalling molecules.
They also have practical lab advantages. Many are chemically stable when lyophilised, amenable to HPLC/MS QC, and compatible with topical vehicles or cell-culture buffers. That said, short half-lives and membrane permeability issues mean you must design assays and formulations with peptide chemistry in mind.
Key peptides used in skin glow, nail integrity, and beard biology—with what they do
Below are peptides you’ll see often in the literature or in preclinical screening. I name mechanisms you can test in vitro or ex vivo, and the common lab endpoints.
GHK‑Cu — copper tripeptide that modulates collagen synthesis, MMP expression, and fibroblast activity. It’s commonly measured for ECM remodelling and wound-healing endpoints. BPC‑157 — a gastric peptide fragment that appears in wound-healing and angiogenesis models; many groups use it to study epithelial restitution and collagen organization in rodents and explants. Thymosin Beta‑4 (TB‑500) — regulates actin dynamics and cell migration; used in tissue-repair models and follicle microenvironment studies. Snap‑8 — short peptide used in topical cosmeceutical assays targeting neuromuscular signalling and expression of proteins involved in expression-related wrinkling endpoints. PE 22–28 and other synthetic fragments — tested for collagen induction, antioxidant response, or melanogenesis modulation in keratinocytes and fibroblasts. Growth-factor mimetics and secretagogues (IGF-1 fragments, GHRPs, etc.) — used in mechanistic work on follicular keratinocytes and dermal papilla cells; pay attention to cross-talk with endocrine pathways in in vivo work.
For practical sourcing and QC, many labs order research-grade GHK‑Cu and multi-component blends to test combinatory effects.

Molecular mechanisms you can assay: collagen, keratins, MMPs, and more
Mechanistically, peptides that influence "glow" tend to converge on extracellular matrix (ECM) turnover, keratinocyte differentiation, barrier function, and local microvascular control. For nails and beard follicles, keratin gene expression, follicle cycling regulators, and matrix interactions matter most.
Common molecular endpoints and assays
Collagen synthesis: hydroxyproline assay, procollagen I/III ELISA, qPCR for COL1A1/COL3A1. MMP activity: zymography, MMP ELISAs, TIMP expression profiling. Keratins: qPCR and immunostaining for KRTs relevant to nails (KRT6, KRT16 family) or hair (KRT31–KRT40 subsets). Cell proliferation and migration: Ki‑67 IHC, BrdU/EdU incorporation, scratch assays, transwell migration. Barrier metrics: transepithelial electrical resistance (TEER) in keratinocyte layers, tape-stripping TEWL in ex vivo skin. Angiogenesis and perfusion surrogates: tube formation assays, CD31 staining in explants, or Doppler flow measures in animal models.
Design the molecular readouts to match your hypothesis. If you think a peptide stabilises collagen, pairing hydroxyproline quantification with MMP zymography gives a clearer picture than either assay alone.
Models and experimental design: cells, explants, and animals
Your model determines which endpoints are realistic. Primary human dermal fibroblasts and keratinocytes are ideal for mechanistic signalling and ECM work. Immortalised lines (HaCaT, NIH/3T3) are cheaper and reproducible, but they differ in differentiation and matrix deposition. Organotypic 3D skin equivalents bring back a stratified epidermis and are excellent for topical vehicle testing and barrier assays.
Ex vivo human skin explants bridge in vitro and in vivo. They maintain architecture and appendages for several days and let you test topical penetration and local immune signalling without systemic confounders. Keep viability limits in mind; explant metabolism drops after 3–7 days depending on conditions.
Rodent in vivo models let you study follicle cycling, angiogenesis, and integrated wound repair. Choose endpoints and power accordingly, and include appropriate vehicle and peptide controls. Randomisation and blinded scoring of histology is non‑negotiable if you want reproducible data.
Delivery and formulation considerations for peptide experiments
Peptides are hydrophilic and can have limited skin penetration. For topical studies you'll pick a vehicle based on the peptide's size and intended target layer. Water‑based gels, solvent mixes (propylene glycol/ethanol blends), and liposomal or nanoparticle carriers are common. For follicle-targeted work, penetration enhancers or microinjection into the pilosebaceous unit are used in experimental settings.
For systemic or subcutaneous experiments in animals, consider stability in serum and potential proteolysis. PEGylation, cyclisation, or use of D‑amino acids can increase half-life, but those chemical changes alter mechanism and must be validated with the same assays you plan to use downstream.
Topical: vehicle selection, occlusion studies, Franz diffusion cells for permeability. Local injection: depot formation, tissue distribution, and local inflammation scoring. Systemic: pharmacokinetics, clearance rates, and potential off-target endocrine effects in vivo.
Stability, handling, and quality control in the lab
Store lyophilised peptides desiccated, usually at −20°C or colder unless the manufacturer specifies otherwise. Once reconstituted, most peptides are unstable at room temperature; aliquot and freeze at −80°C when possible to avoid freeze–thaw cycles. Use sterile, nuclease-free solvents for reconstitution if you'll apply peptides to cells or explants.
QC matters. HPLC purity and MALDI/ESI mass spec verification are typical. Check for truncation peaks, dimers, and oxidation products—methionine and tryptophan residues are common trouble spots. Acceptance criteria should match your experimental sensitivity: for signalling assays you might accept 90% purity; for in vivo work you’ll usually want >95% and clear identity confirmation by MS.
Assays specific to nails and beard follicles
Nail biology is often overlooked, but it’s quite distinct from skin. Nails are composed of hard keratins with different expression patterns than epidermal keratins. For nails, useful readouts include keratin-expression panels, nail plate tensile testing, and ex vivo nail organ culture where you can measure growth rate and protein composition.
Beard and scalp follicles are similar but not identical. Dermal papilla cell cultures and hair‑follicle organ culture allow direct observation of anagen versus catagen induction. Quantitative endpoints include follicle length, hair-shaft diameter, and proliferation markers in the bulb. In rodents you can score hair-cycle stage histologically or use phototrichograms to quantify density changes over time.
Data interpretation: common pitfalls and how to avoid them
Expectation bias is common in cosmetic peptide research. Small subjective improvements—glow, sheen, perceived density—often show up in uncontrolled human work. In the lab, make endpoints objective: blinded image analysis, automated collagen quantification, or peptide‑tagged reporters where possible.
Also watch out for cell-culture artefacts. High peptide concentrations can be cytotoxic or trigger non‑physiological stress responses. Run cell viability assays alongside mechanistic readouts and use concentration ranges spanning an order of magnitude. If you move to explants or animals, check peptide stability in the chosen matrix; a peptide stable in buffer may be cleaved rapidly in serum or by skin proteases.
Control for vehicle effects: many solvents alter barrier function or cell phenotype. Verify target engagement: show changes in downstream signalling, not only the clinical endpoint. Replicate across donors for explant work—human skin is highly variable.
Practical resources: assays, kits, and peptide products for lab work
When planning a study, assemble your assay suite before you order large peptide lots. Hydroxyproline kits, MMP zymography reagents, and validated antibodies for collagen I/III and hair‑keratin subtypes save time. If you need a starting panel of research-grade peptides for screening, suppliers offer single compounds and blended formulations for combinatory testing.
Keep procurement aligned with your QC requirements. Ask for certificates of analysis showing HPLC traces and mass spec identity, and verify storage recommendations for each peptide. For topical formulation work, small‑scale compounding and Franz cell permeability studies will tell you whether a peptide reaches the desired stratum.
We stock several peptides commonly used in skin and appendage research as research-only products. Check each product’s COA and stability notes before use, and remember all work should be limited to lab research models.
Ethics, regulatory, and reporting notes for publication-ready work
Label everything as research-only in methods and supplier sections. For animal work, follow institutional IACUC protocols and report randomisation, blinding, and sample-size calculations in the methods. For ex vivo human tissue, include IRB approval and donor demographics because age and anatomical site heavily influence skin and hair outcomes.
Finally, be transparent about peptide provenance and QC in manuscripts. Report purity, solvent for reconstitution, storage conditions, and any modifications such as PEGylation or cyclisation. Reproducibility lives in those details.
Peptides are powerful experimental tools for studying ECM remodelling, keratin biology, and follicle dynamics—if you match the peptide chemistry to the right model and the right readouts. Keep assays objective, QC tight, and reporting detailed, and you’ll get data that move the science forward.